EU regulator recommends adding heavy periods to side effects of mRNA COVID shots
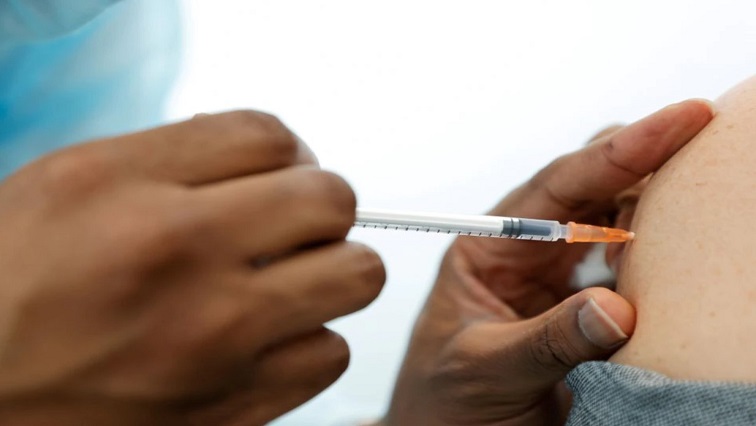
A European Medicines Agency (EMA) committee on Friday recommended adding heavy menstrual bleeding to
EU regulator backs wider use of AstraZeneca COVID therapy

Europe’s medicines regulator has backed using AstraZeneca’s preventative COVID-19 therapy as a
Europe considers “dose-sparing” to increase monkeypox vaccine, WHO seeks trials
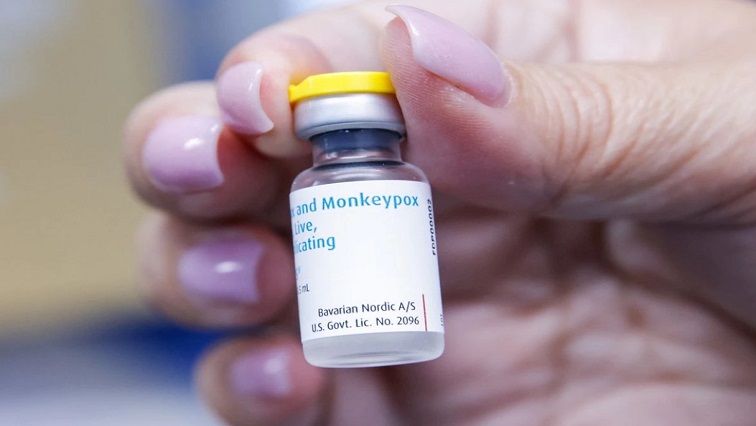
Health officials in Europe are discussing whether to follow a move by the United States to stretch
Benefits outweigh the risks of AstraZeneca COVID-19 shot as review continues: WHO

A World Health Organisation (WHO) vaccine safety panel said on Wednesday that it considers that the
Benefits of AstraZeneca outweigh risks: EMA

The head of the European Medicines Agency, EMA, Emma Cooke has reiterated that there is no
EMA committee recommends restrictions on fluoroquinolone, quinolone antibiotics

The European Medicines Agency’s risk assessment committee has recommended restrictions on the use of

