The South African Medical Research Council (SAMRC) says they have strengthened their vaccine safety monitoring in line with the South African Health Products Regulatory Authority (SAHPRA) regulations.
The first batch of Pfizer’s vaccines arrived in South Africa on Sunday night.
More than 325 000 doses landed at OR Tambo International Airport and they were transported to a central warehouse under strict security measures.
First batch of Pfizer vaccines arrives in SA:
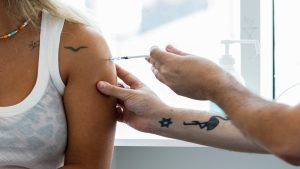
So far more than 18 000 health workers have been vaccinated under the Sisonke protocol and have received the Johnson & Johnson vaccine.
SAMRC chair, Professor Glenda Gray, says people should report persistent side-effects after inoculation.
“We have increased our safety reporting to SAHPRA as well as training the staff at the site. We also lookout for any rare cutting disorders. These vaccines are very reactive genic whether Pfizer or the J&J. Obviously, we expect lot of side effects at the time of vaccination. ”
“But if people who take the vaccine, after day 4 if they have an unrelenting headache or unexplained rear abdominal pain they should contact the safety desk to report this,” explains Gray.
Below is the full interview with Prof Gray:
The video below looks at the Pfizer COVID-19 batch: